 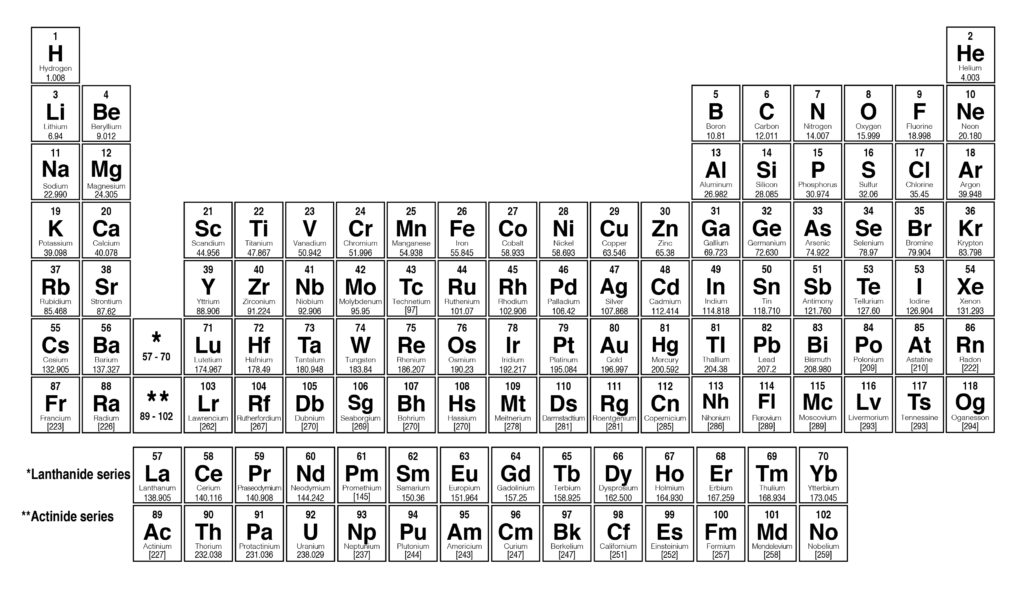 
By following the standard notation, you can avoid ambiguity in parsing and obtain accurate results.). For example, C2H5OH for ethanol, H2O for water, or Na2SO4 for sodium sulfate. 1 1.007 94 H Hydrogen 3 6.941 Li Lithium 11 22.989 77 Na Sodium 19 39.0983 K Potassium 37 85.4678 Rb Rubidium 55 132.905 45 Cs Cesium 87 (223) Fr Francium 4 9. It is important to ensure that the chemical formula is entered correctly using standard notation. 2: Acetic acid (left) has a molecular formula of C2H4O2 C 2 H 4 O 2, while glucose (right) has a molecular formula of. They are very different compounds, yet both have the same empirical formula of CH2O CH 2 O. The structures of both molecules are shown in the figure below. These three pieces of data are the elemental symbol, the atomic number (typically given the symbol, Z, and the atomic weight. Its molecular formula is C6H12O6 C 6 H 12 O 6. 
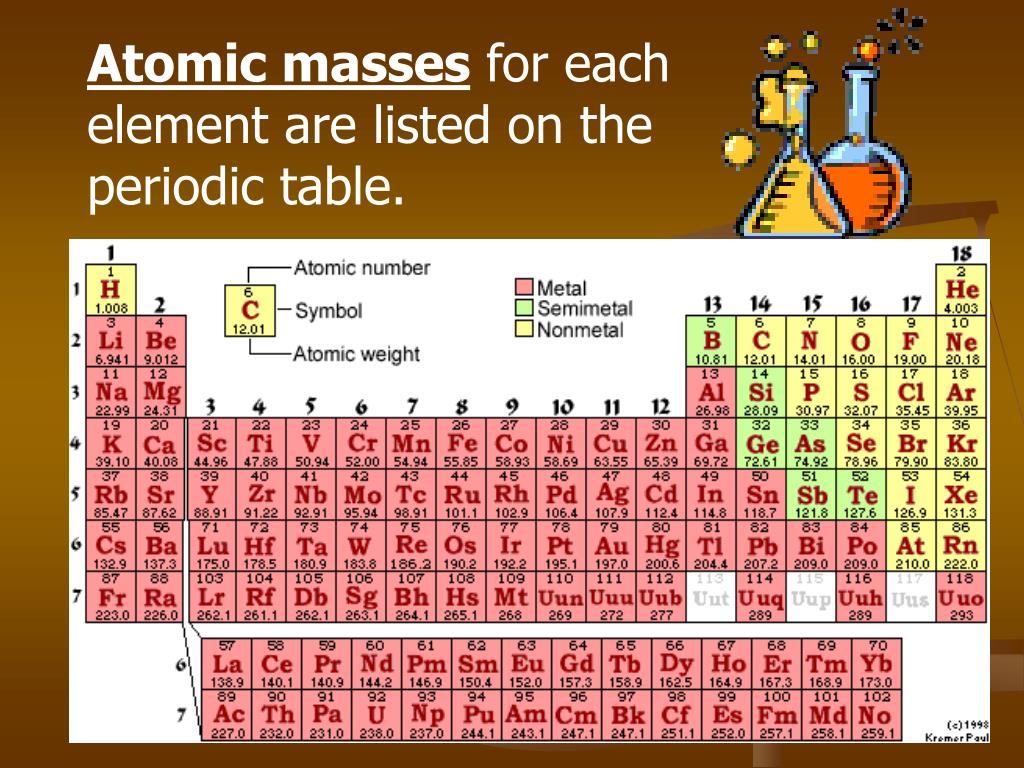
In it's simplest form (shown below), each entry only has three pieces of information that you will need to know. The atomic mass is the average number of protons and neutrons in atoms of a chemical elements, allowing for the natural abundances of the element’s isotopes. Core share and HTML view are not possible as. 1 Orientation: what is physical chemistry about. The result table includes the element symbol, atomic mass, multiplier (indicating the number of atoms of each element in the formula), and the calculated result. The periodic table can often be presented with an abundance of data about each and every element listed. This is a periodic table with atomic mass, element name, element symbol, and atomic number. Appendix F - Periodic table of the elements, with molar masses. In addition to the molar mass, the calculator also provides a result table with detailed information on the individual elements in the formula. It then calculates the molar mass by adding up the atomic weights of all the elements in the substance. The calculator utilizes the periodic table of elements to determine the atomic weights of the elements present in the formula. Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygens is 16. To use the calculator, simply input the chemical formula of the substance and click the "Calculate" button. The Periodic Table of the Elements 1 H Hydrogen 1.00794 2 He Helium 4.003 3 Li Lithium 6.941 4 Be Beryllium 9.012182 5 B Boron 10.811 6 C Carbon 12.0107 7 N Nitrogen 14.00674 8 O Oxygen 15.9994 9 F Fluorine 18.9984032 10 Ne Neon 20.1797 11 Na Sodium 22.989770 12 Mg Magnesium 24.3050 13 Al Aluminum 26.981538 14 Si Silicon 28. However, if we have 6. This number is usually expressed as a decimal rather than as a whole. According to the periodic table, the atomic mass of aluminum is 26.98 amu, copper is 63.55 amu, and carbon is 12.01 amu.Since 1 amu is only 1.674 × 10 24 g, these masses would be way too small to measure on ordinary laboratory equipment. This is almost always written as a number at the bottom of the element's square on the table, under its one or two letter chemical symbol. Most standard periodic tables list the relative atomic masses (atomic weights) of each element. Molar mass is the mass of one mole of a substance and is expressed in grams per mole. Locate atomic mass on the periodic table. You can search for any element by its symbol, name, or atomic number, and see its molar mass, weight, energy levels, electronegativity, and more. The Molar Mass Calculator is a tool used to calculate the molar mass of a substance based on its chemical formula. Ptable is a comprehensive online resource for the periodic table of the chemical elements, with detailed information on their properties, isotopes, isotopes, and isotopes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
